Academic pharmacist Nataly Martini discusses the medical management of asthma in adults and adolescents, which has evolved to prioritise early anti-inflammatory treatment. She also explains how to improve patient outcomes by proactively identifying poor asthma control and supporting equitable access to education and treatment
Adverse events following immunisation with COVID-19 vaccines: Safety Report #1 – 6 March 2021
Adverse events following immunisation with COVID-19 vaccines: Safety Report #1 – 6 March 2021
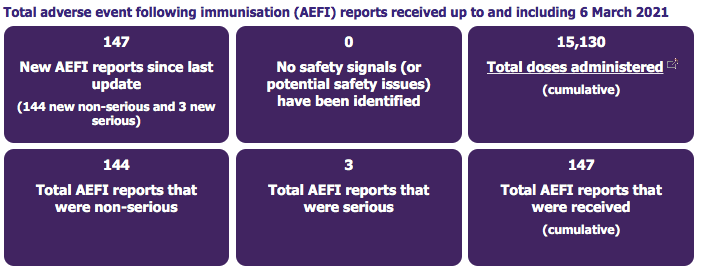
This page [report] provides information on the number of adverse events following immunisation (AEFI) reports received for COVID-19 vaccines.
The national roll-out of COVID-19 vaccines commenced on 20 February 2021. The vaccine currently available in New Zealand is Pfizer-BioNTech (Comirnaty). All medicines can cause side effects, the known side effects for Comirnaty are listed in the data sheet and consumer medicine information.
Suspected AEFI to COVID-19 vaccines are reported to the Centre for Adverse Reactions Monitoring (CARM). The Ministry of Health (through Medsafe) contracts the collection of this information to CARM, based at the University of Otago in Dunedin. Medsafe is closely monitoring the AEFI reported from the use of the COVID-19 vaccine. Find out more about vaccine safety monitoring
The three serious cases were all allergic reactions and all managed appropriately.
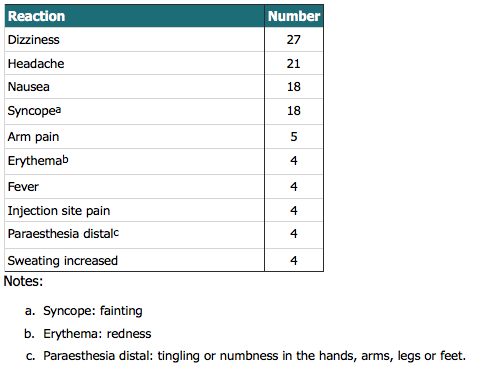
Please note that one adverse event report, which represents one person, may report on more than one symptom. Reports are sent to CARM if the reporter suspects that the vaccine may have caused the event. This does not necessarily mean that the vaccine did cause the event.
The number of reports can be influenced by how many people are being vaccinated, media attention, the nature of the events (eg, how painful the vaccination was), and other factors which vary over time. Not everyone who has an adverse reaction reports it, and some people may report AEFIs after each vaccination. The information here shows the number of reports not the number of people who experienced an AEFI.
The information is limited by the information provided in the report and may change over time due to quality control procedures and/or receipt of additional information. Non-valid reports are not included in the data.
Adverse event following immunisation (AEFI)
An AEFI is an untoward medical event which follows immunisation and does not necessarily have a causal relationship with the administration of the vaccine. The adverse event may be an unfavourable or unintended sign, abnormal laboratory finding, symptom or disease.
Serious adverse event following immunisation
An AEFI is considered serious if it:
- is a medically important event or reaction
- requires hospitalisation or prolongs an existing hospitalisation
- causes persistent or significant disability or incapacity
- is life threatening
- causes a congenital anomaly/birth defect
- results in death.
It is possible for different people to have experienced the same event but for the report to be serious for one person and non-serious for another person.
Safety signal
Information on a new or known adverse event that may be caused by the vaccine and requires further investigation. Safety signals can be detected from a wide range of sources such as CARM reports, clinical studies and scientific literature.
More information
See the data sheets and consumer medicine information for the expected reactions for approved COVID-19 vaccines.
COVID-19 Vaccine Safety Monitoring Process
Medsafe and CARM thank everyone who has contributed to the monitoring of COVID-19 vaccines. Please continue to report any adverse events following immunisation.