Academic pharmacist Nataly Martini discusses the medical management of asthma in adults and adolescents, which has evolved to prioritise early anti-inflammatory treatment. She also explains how to improve patient outcomes by proactively identifying poor asthma control and supporting equitable access to education and treatment
CLASStime: Dry eye disease
CLASStime: Dry eye disease

Dry eye disease affects a large number of people and causes a significant degree of morbidity. It can usually never be cured, but good management of the underlying causes can allow significant improvements in quality of life. Nick Mathew provides an overview of this common condition and its management in primary and secondary care
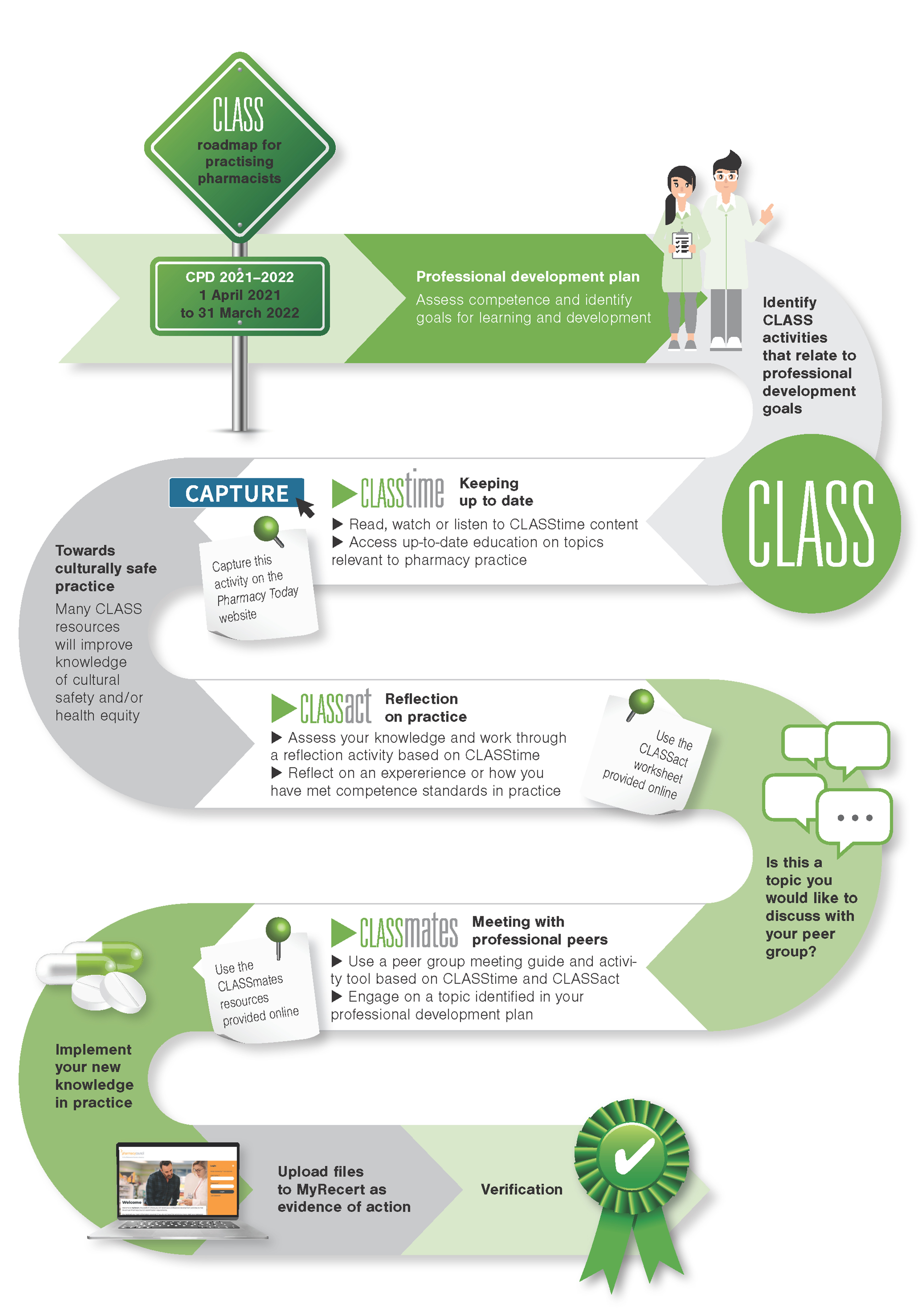
Our suite of CLASS activities provides meaningful independent and peer group learning that you can put into action, while helping you complete your annual recertification requirements.
You can choose how many elements you wish to undertake, depending on your professional development plan, but it is recommended that you complete both CLASStime and CLASSact before CLASSmates. A new topic will be covered each month.

This CLASStime article supports professional development with respect to:
- Understanding the aetiology and pathogenesis of dry eye disease
- Identifying risk factors and symptoms associated with DED
- Counselling patients on effective strategies for managing DED
As you read, highlight new knowledge or areas where you need to know more. You can also use the Capture button online to keep notes of your thoughts and reflections on this article. This may be useful if you choose to proceed to CLASSact and CLASSmates for this topic.
Completing this CLASStime activity may allow you to fulfil some or all of the following elements of your annual recertification requirements:
- Keeping up to date – go to MyRecert (myrecert.pharmacycouncil.org.nz), select the “Keeping up to date” page of your portfolio, then enter a description of this article
- Reflection on practice
This activity has been endorsed by the Pharmaceutical Society of New Zealand as suitable for inclusion in a pharmacist's continuing education records for continuing professional development purposes.
- Dry eye disease often has more than one cause and is best treated by targeting multiple aspects of the problem.
- Left untreated, DED can lead to significant morbidity.
- Tear quality is as important as tear quantity.
- Inflammation of the ocular surface is a key factor in both the development of DED and damage caused by DED.
Dry eye disease is a common cause of irritable red eyes and affects a large percentage of the adult population. It reduces quality of life and is usually chronic. There is no “magic bullet” to treat dry eye disease, but increased awareness of the causes and targeted treatment of the underlying conditions has led to better management.
Dry eye disease (DED) is not solely due to a lack of tears; it can be caused by damage to, or a deficiency of, any part of the complex system that maintains the ocular surface. Two reports from the International Dry Eye Workshop (DEWS in 2007 and DEWS II in 2017) have been instrumental in helping improve the understanding of DED and focusing treatment towards the underlying pathology. The latest report gave us an updated definition of DED:
“Dry eye is a multifactorial disease of the ocular surface characterised by a loss of homeostasis of the tear film, and accompanied by ocular symptoms, in which tear film instability and hyperosmolarity, ocular surface inflammation and damage, and neurosensory abnormalities play etiological roles.”1
The tear film has multiple purposes, all of which are equally important:
- lubrication and nourishment of the eye surface
- removal of microbes and foreign material
- a means for the immune system to access the surface of the eye
- provision of a smooth optical surface for clear and sharp vision.
The tear film is key to the regeneration and healing of the ocular surface, and its components need to be in balance. Tears are not purely an aqueous fluid but a mixture of key components that combine into a tear film, which is primarily lipid on the surface, covering a mucoaqueous layer beneath.
The lipid layer – wax and cholesteryl esters (non-polar lipids) make up the majority of the lipid layer and are spread onto the mucoaqueous layer by an underlying layer of polar lipids, including (O-acyl)-omega-hydroxy fatty acids and possibly phospholipids.
The lipid layer is produced by modified sebaceous glands, called meibomian glands, along the upper and lower lid margins. Inflammation of these glands is an important and common factor in many cases of DED.
The oily secretion (meibum) helps improve tear quality and stability, and reduces tear evaporation. However, the tear film lipid layer is just one part of the overall mechanism that keeps the tear layer stable. It is thought that interactions of the whole tear film and eyelid ecosystem, including lipids, mucins, proteins and salts, work to prevent evaporation and tear film collapse.
The mucoaqueous layer – mucin is secreted from the conjunctival goblet cells and helps improve the wetting of the surface of the eye and cornea. The lacrimal gland produces the aqueous component, which contains electrolytes, important antibacterial proteins and immunoglobulins. The aqueous layer combines with the mucin layer and supplies oxygen and electrolytes to the surface of the eye.
The condition and function of the eyelids is also very important – if the tear film is not spread evenly and frequently across the eye, it is unable to perform its functions. Blinking spreads the tear film over the eye towards the medial canthus. Tears are then drawn into the superior and inferior puncta (one punctum in each eyelid) to enter the lacrimal canaliculi, and from there to the nasolacrimal duct, which drains into the nose.
Aqueous-deficient dry eye – the less common category. It can arise from autoimmune disorders, such as Sjögren syndrome where activated T-cells infiltrate the lacrimal and salivary glands, reducing their function. It is possible, but uncommon, to develop aqueous deficiency due to lacrimal gland damage.
Evaporative dry eye – the majority of DED is due to excessive tear evaporation. The underlying cause of this loss is often poor tear quality secondary to meibomian gland dysfunction (Figure 2). Poor meibomian gland condition leads to inflammation and causes burning, sore and gritty eyes. Lack of a good-quality lipid layer leads to a poor-quality tear film, ocular surface damage and further inflammation, which will, in turn, lead to further deterioration of the meibomian glands.
Thus, treating lid inflammation is key to improving ocular surface health and dry eye symptoms.
Reduced aqueous tear volume and increased evaporation of the aqueous components of the tear film both lead to tear hyperosmolarity, a key step in the development of DED. This gives rise to cell death and triggers inflammation, which further damages the ocular surface and leads to loss of mucin-producing goblet cells. This, again, exacerbates tear film instability and drives the cycle of events that leads to further ocular surface inflammation.
Chronic lid margin inflammation also causes rough, irregular lid margins (Figure 3) that lead to ocular surface problems later in life. It is important to address these problems early on to avoid chronic and irreparable damage to the lid margin and ocular surface.
Ocular rosacea is a more extreme presentation of meibomian gland dysfunction with more severe symptoms of erythema and swelling of the eyelids. It can lead to corneal neovascularization and scarring.
Abnormalities of lid closure from exophthalmos, nerve damage or lid trauma can cause severe dry eye problems from ocular surface exposure.
Older people and women are more likely to experience DED, but there are several other important risk factors.
Environmental – low humidity is a contributing factor, especially exposure to dry, windy conditions. Working or playing sport outdoors may exacerbate dry eye problems.
Occupational – computer use and concentrated tasks lead to reduced blinking, which increases tear evaporation and can lead to poor spreading of the tear film across the eye surface.
Nutritional – some diets too high in omega-6 and too low in omega-3 fatty acids can contribute to DED, as can vitamin A deficiency.
Hormonal – androgen deficiency has been promoted as a cause of meibomian gland dysfunction and evaporative tear loss. However, postmenopausal hormone replacement therapy (oestrogen alone more so than combined HRT) has also been associated with an increased risk of DED.
Systemic medications – many systemic medications can cause DED, usually by aqueous suppression. Any change in medication should be considered as a possible contributing factor. Classes of systemic medication known to cause DED include anticholinergics, beta-blockers, diuretics, amiodarone and isotretinoin. Isotretinoin stops the normal function of the meibomian glands, causing a severe lipid layer deficiency and chronic DED that takes years to resolve, if at all.
Topical ophthalmic medications – any topical eye drop that is preserved with benzalkonium chloride can lead to ocular surface problems contributing to, or easily confused with, DED. The active components of many drops can also cause eye irritation, particularly glaucoma medications – prostaglandin analogues, brimonidine and iopidine. It is possible to develop allergic reactions months to years after starting some eye drops.
Contact lens wear – use of contact lenses disrupts the tear film and, in some cases, blinking. Any person with DED who wears contact lenses should have a thorough review of the type of contact lens they wear, the care system (solutions) they use and the surface of the eye.
Refractive surgery – LASIK vision correction surgery disrupts corneal innervation and can contribute to aqueous deficiency. Temporary dryness is a common feature of LASIK in the first six to 12 weeks post-operatively, but it can become chronic in rare cases, particularly if a person is already predisposed to dry eyes. If this occurs in the early post-operative period, it should be managed in consultation with the surgeon.
Other conditions – reduced blink rate is a common feature of Parkinson disease, resulting in increased evaporation. Other important conditions that are associated with dry eyes are diabetes mellitus and autoimmune diseases.
A patient’s symptoms (Panel 1) are a key part of the diagnosis of dry eye. Most symptoms of DED occur in both eyes. Symptoms in one eye are unlikely to be DED unless a contributing factor affects only that eye.
No single test can be used to diagnose DED. The most important considerations are the patient’s symptoms and the condition of the eyelids, lid margin, cornea and conjunctiva. General gross examination can detect lid abnormalities and lid margin inflammation.
Redness in DED is typically diffuse but can be more concentrated at the three and nine o’clock areas of the bulbar conjunctiva near the limbus (areas that are more exposed between blinks).
Tear quality and quantity is difficult to judge without a slit-lamp microscope or tear analysis devices, but excessive blinking can be a sign.
Slit-lamp biomicroscopy is an important investigation and is the main tool for accurate diagnosis of DED. This is sometimes available in general practice but universally in ophthalmology and optometry. In a specialist ophthalmic practice, there are many other tests available to help grade severity and investigate tear quality and potential problem areas.
- Dry, gritty, sore eyes.
- Long-term foreign body sensation – “it feels like sand in my eyes”.
- Stinging.
- Burning – often associated with ocular surface or lid margin inflammation.
- Itching – more often associated with allergic eye problems, but this term may be used by some people for symptoms related to dryness.
- Smeared or blurry vision that fluctuates with blinking.
- Light sensitivity – for example, sore and watery eyes in bright sunlight.
- Red eyes.
- Not being able to wear contact lenses for the durations worn previously.
- Intermittent paradoxical watering (eg, in dry, windy conditions) – this is reflex tearing in response to an irritated cornea, which can happen if the eyes are dry, and is a more common cause of watery eyes than blocked tear drainage.
The mainstay of dry eye treatment is still the use of lubricating drops (tear supplements). However, increased understanding of the role of ocular surface inflammation means short-term topical steroids are becoming more widely prescribed. The side effects of these require careful monitoring.
Most patients have more than one cause for their problems and benefit from treatment of as many aspects as possible. Treatment should generally begin with the least invasive and simplest options, progressing from there.
Environment
The importance of the person’s environment should not be discounted. This does not mean changing jobs or lifestyle, but making simple modifications, such as moving away from office air conditioning vents or having short breaks every hour when computer use predominates. Sunglasses are also very helpful in limiting ultraviolet damage to the structure of the eyelid, and they help minimise chronic damage for people who work outdoors.
Lid hygiene and massage
Regular hot compresses and gentle massage of the lid margins are helpful for improving meibomian gland insufficiency and mild blepharitis (see Panel 2 for advice to patients). Special devices (eg, LipiFlow) are more effective than hot compresses and lid hygiene, but these are less accessible and typically only used by specialist practices.
For patients with seborrheic blepharitis, washing the eyes daily and removing dry crusts from the lashes also helps reduce lid and ocular surface inflammation.
Dietary advice
Omega-3 fatty acids have been shown to have anti-inflammatory effects, and there is good evidence of their usefulness in DED. Increased omega-3 intake can be achieved by diet adjustment or commercial supplements. Foods rich in omega-3s are:
- walnuts
- wild rice
- beans (in particular, black beans and kidney beans)
- deep-sea, cold-water fish (eg, salmon, sardines, mackerel)
- broccoli, spinach, pak choi, kale, cauliflower, onions
- canola oil (for cooking) and flaxseed oil (salad oil/dressing).
- Warm the outside of the eyes with a hot washcloth for one to two minutes, or close the eyes under a warm shower.
- Dry the face and, with a dry finger, apply firm pressure on the edge of the lower eyelid at the base of the eyelashes (this should not cause any discomfort). Hold for a few seconds.
- Repeat the application of pressure in four or five places along the lower lid.
- The upper lid can be massaged by looking down, closing the eyes, and pressing in and down gently. Upper lid massage is less effective than lower lid massage due to the anatomy of the eyelid.
Supplemental artificial tears are useful in all dry eye problems.
Tear supplements
Supplemental artificial tears are useful in all dry eye problems. Artificial tears usually contain much more than just an aqueous supplement. Most contain electrolytes and some form of viscosity agent, such as hydroxypropyl methylcellulose (hypromellose) or sodium hyaluronate.
Due to our increased understanding of tear physiology, we can now make recommendations for products that will target specific tear film components and different subtypes of DED. For example, supplements that mimic the lipid layer may work better in cases of evaporative dry eye and meibomian gland dysfunction.
Usually, the progression of treatment depends on severity and begins with lower-viscosity, preserved drops. A more viscous tear supplement will last longer on the eye, but even mildly viscous drops blur vision on application. This can last from half a minute to an hour or more.
A very oily drop such as Refresh Night Time (containing soft white paraffin) can still cause blurred vision the next morning, but it is very effective for severe dry eyes and problems related to corneal exposure. Poly Gel and Viscotears Gel are less blurry to use.
There is also the option of preserved and non-preserved drops. Modern preservatives used in artificial tears are less likely to cause topical sensitivity than in the past, but some sensitivity to preservatives still occurs, especially with frequent use. Thus, more severe dry eye may require preservative-free drops. These are usually supplied in small ampoules, which contain enough for either a single application or a single day’s use.
There are also preservative-free, multi-dose bottles that have special one-way dispensers to prevent contamination – Hylo-Fresh is a good example of this. Systane Ultra is also available as a preservative-free drop in a multi-dose bottle.
Tear supplements can be prescribed, relieving some of the ongoing financial burden of treatment. In some circumstances, Special Authority can be granted for preservative-free drops (Hylo-Fresh multi-dose bottle, Systane single-dose units, Poly Gel single-dose units) on application by any relevant practitioner using form SA1388.
The drops should be used to prevent dryness…this is more effective than just using them for symptomatic relief
The best advice to give a patient using tear supplements is that the drops should be used to prevent dryness, and that this is more effective than just using them for symptomatic relief. For example, if the eyes frequently feel dry at the end of the day, use tear supplements once or twice in the mid-morning.
Punctal plugs
Retention of the aqueous tears is a useful approach in some cases. Punctal plugs can be temporary or semi-permanent and are generally placed in the lower (sometimes also the upper) canaliculus to limit tear drainage. These can help retain tears in aqueous-deficient DED but are less helpful in cases of ocular surface inflammation as they cause stagnation of the tears and increased tear osmolarity.
Topical corticosteroids
As mentioned, ocular inflammation and meibomian gland dysfunction are intertwined, and the importance of treating lid inflammation has become widely recognised. Improved meibomian gland function can be gained from some of the treatments above, but inflammation responds best to topical corticosteroids. These can be very helpful in breaking the cycle of inflammation, but even the weakest steroids can have significant side effects.
Risks of topical ophthalmic corticosteroids include epithelial toxicity, crystalline keratopathy, orbital fat atrophy, ptosis, limitation of ocular movement, reduction in endogenous cortisol, raised intraocular pressure and cataracts. While cataracts can be corrected at any stage, glaucoma damage to the optic nerve is irreversible. Once the damage process to the optic nerve has started, it may continue to progress independently of the eye pressure.
Typically, the risks are highest with long-term corticosteroid use, which can be a problem with DED due to its chronic nature – patients are likely to seek out steroids and could use them unsupervised for long periods of time. It should be made very clear to the patient that steroids are for short-term use only.
Inappropriate use of topical corticosteroid in the presence of corneal infection is also a significant problem. The symptoms of an early herpetic keratitis, for example, can be similar to DED, but corneal infections can become blinding if steroids are used. Topical steroids should be prescribed with ophthalmic or optometric supervision.
Steroid-sparing medications are available (eg, topical cyclosporine) but are typically only prescribed by ophthalmologists and specialists in ocular surface problems.
Antibiotics
A useful class of medications for treating meibomian gland inflammation are the oral tetracyclines, typically doxycycline or minocycline. They are also used to treat acne and rosacea, and are relatively safe. They have been shown to be effective at improving irritation symptoms and objectively proven to increase tear film stability. A typical dosage for meibomian gland dysfunction is 50mg or 100mg daily for six weeks.
Taking oral tetracyclines at bedtime or without fluids is a common cause of oesophagitis. Advise patients to take the tablets with food or a large glass of water, and to sit upright or stand for at least 30 minutes afterwards, to help prevent this adverse effect. Oral tetracyclines are contraindicated for children and pregnant women.
Oral azithromycin has been shown to be as effective as, and better tolerated than, the tetracyclines. The risks of QT prolongation should be considered in predisposed patients and in those taking antiarrhythmics, antipsychotic agents, antidepressants or fluoroquinolones. Azithromycin is usually given as a divided dose to reduce adverse gastrointestinal effects (500mg stat then 250mg/day for three days).
The chronic nature of DED means regular follow-up is recommended. This should be multidisciplinary, involving GPs, ophthalmologists and optometrists, who can then send the patient back to the pharmacist with an appropriate management plan.
It is helpful to educate patients about the causes of dry eye – for example, if they understand how meibomian gland dysfunction affects the tear film, then they are more likely to be compliant with lid margin massage and lid hygiene.
The ongoing cost of managing DED should not be underestimated. Depending on the type and brand of eye drop used, costs can easily be several hundred dollars a year. There are funded options that work for most patients, but some types (eg, Hylo-Fresh) require a Special Authority. Once this is in place, any practitioner can supply repeat prescriptions.
Nick Mathew is a therapeutic optometrist at Re:Vision Laser and Cataract in Auckland
Baudouin C, Aragona P, Messmer EM, et al. Role of hyperosmolarity in the pathogenesis and management of dry eye disease: proceedings of the OCEAN group meeting. Ocul Surf 2013;11(4):246–58.
Baudouin C, Aragona P, Van Setten G, et al. Diagnosing the severity of dry eye: a clear and practical algorithm. Br J Ophthalmol 2014;98(9):1168–76.
Blackie CA, Coleman CA, Holland EJ. The sustained effect (12 months) of a single-dose vectored thermal pulsation procedure for meibomian gland dysfunction and evaporative dry eye. Clin Ophthalmol 2016;10:1385–96.
Perry HD. Dry eye disease: pathophysiology, classification, and diagnosis. Am J Manag Care 2008;14(3 Suppl):S79–87.
McGhee CN, Dean S, Danesh-Meyer H. Locally administered ocular corticosteroids: benefits and risks. Drug Saf 2002;25(1):33–55.
Foulks GN, Borchman D, Yappert M, et al. Topical azithromycin and oral doxycycline therapy of meibomian gland dysfunction: a comparative clinical and spectroscopic pilot study. Cornea 2013;32(1):44–53.

Please CAPTURE this article before leaving this page. To move on to CLASSact, where you can download an interactive PDF that provides you with both knowledge and reflective questions for independent learning, click the button below.
Completing this CLASSact activity may allow you to fulfil some or all of the following elements of your annual recertification requirements:
- Keeping up to date
- Reflection on practice


Please CAPTURE this article before leaving this page. To move on to CLASSmates, where you can download a peer group meeting guide and activity tool based on CLASStime and CLASSact, click the button below. It includes a facilitator guide, user guide, notes template and Jamboard tool (for online meetings).
Completing the CLASSmates activity may allow you to fulfil some or all of the following elements of your annual recertification requirements:
- Keeping up to date
- Reflection on practice
- Meeting with professional peers

- Craig JP, Nelson JD, Azar DT, et al. TFOS DEWS II report executive summary. Ocul Surf 2017;15(4):802–12. www.tfosdewsreport.org